Parasite Infection (Leishmaniasis) in Dogs
Introduction
Leishmaniasis is a severe, widespread zoonotic disease which occurs in the Middle and Far East, the Meditterranean basin, South America, and in some states in the United States of America. Leishmania is primarily a parasite of rodents, carnivores, marsupials, edentates, insectivores, and secondarily of dogs and humans. Severe disease develops in the man and in the dog, which is characterized either by skin lesions or a general visceral involvement. The dog is the reservoir for visceral leishmaniasis in South America and the Meditteranean region. The disease is transmitted by the blood sucking sandfly (genus Phlebotomus in the Old World and genus Lutzomya in the New World). The disease is caused by several species of protozoa in the genus Leishmania. Visceral forms of leishmania are caused by L.donovani and L.infantum/L.chagasi in the Old World and L.infantum/L.chagasi in the New World.
What are the signs and symptoms of leishmaniasis in dogs?
Some dogs can have the Leishmania parasite for extended periods of time and may not exhibit any signs or symptoms of disease (asymptomatic). In asymptomatic dogs, the parasite can lay dormant for a period of time, sometimes years, before a stimulant, such as stress or illness, triggers the parasite to multiply and attack the body and eventually lead to cutaneous leishmaniasis or visceral leishmaniasis. However, both asymptomatic and symptomatic dogs are capable of infecting sand flies and spreading the disease.
In most symptomatic dogs, the first sign of disease appears about 2-4 months after the initial infection. Symptoms may include sores on the skin, peeling, ulcers, loss of weight, bald patches, conjunctivitis, blindness, nasal discharge, muscular atrophy, inflammation, swelling, and organ failure, including mild heart attacks.
How do I prevent my dog from getting leishmaniasis?
There is currently no medicine to prevent leishmaniasis in dogs. The best way to prevent your dog from getting infected is to avoid regions of the world where it is found. Additionally, do not allow your dogs near a dog that is suspected of having the infection.
What should I do if I think my dog has leishmaniasis?
Contact your veterinarian. More information on leishmaniasis in dogs can be found at http://www.capcvet.org/recommendations/leishmaniasis.html
Can I get leishmaniasis from my dog?
No. There have been no documented cases of leishmaniasis transmission from dogs to humans.
Etiology
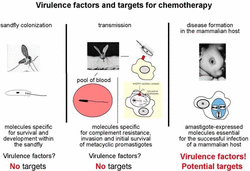
Leishmaniasis is a group of infections caused by protozoan of the genus Leishmania. There are a variety of clinical forms. In nature all existing species of Leishmania are transmitted to humans and other mammals by the bite of infected females sandflies. Invertebrate hosts are restricted to species of hematophagous Phlebotomus (Order Diptera, Family Psychodidae, Sub-Family Phlebotominae), especially subspecies Lutzomyia longipalpis in the New World and genus Phlebotomus in the New World.
In the Americas, human leishmaniasis can be divided in two broad categories: cutaneous and visceral leishmaniasis. Cutaneous leishmaniasis has a large variety of forms which can be grouped in the following manner: cutaneous leishmaniasis, characterized by localized skin lesions which can heal spontaneously or become chronic lesions with disfiguring scars; mucocutaneous leishmaniasis, characterized by ulcerative lesions and destruction of the mucosa; diffuse cutaneous leishmaniasis, characterized by nodular lesions which are not ulcerative and disseminated. The visceral form of the disease is chronic and progressive and affects various organs, including spleen, liver, bone marrow, lymph nodes and skin.
The cycle of Leishmania involves a vertebrate host and Phlebotomus fly as a vector. In the mammalian host, Leishmania is an obligate intracellular parasite and exists in the amastigote form inside cells of the mononuclear phagocytic system.
The amastigote forms are characterized as circular, about 5 microns in diameter, having a nucleus, kinetoplast and rudimentary flagellum. It multiplies by binary fission, repeatedly until the host cell is destroyed. Inside the gastrointestinal tract of the sandflies, the amastigote transforms into paramastigotes and promastigotes – elongated, flagellated, motile forms, which have a central nucleus and terminal kinetoplast.
The transmission of the disease to vertebrate hosts occurs predominantly by inoculation of the infective promastigote form during a bite of the sandfly. However, other possible routes included direct contact, transplacental, venereal and blood transfusions.
After inoculation in mammalian hosts, the infective promastigote attaches to macrophages through different cellular receptors, and it is then localized into a vacuole which fuses with lysosomes The parasite survives phagocytosis and undergoes different metabolic transformations, becoming amastigote forms, which lyse the host cells and then infect other phagocytic mononuclear cells.
Visceral leishmaniasis in humans is known as kala-azar, ,which means lethal disease in Hindu. In the Old World the etiologic agents are Leishmania donovani and Leishmania infantum and in the New World, Leishmania chagasi.
Epidemiology
Visceral leishmaniasis is currently considered an emerging and reemerging disease in both rural and urban areas. According to the World Health Organization (WHO), in 1990 there were 12 million cases with 400 thousand new cases every year. In Brazil, the estimate is approximately 3000 cases per year.
The WHO has currently pointed out the increase in the number of cases of co-infection HIV/visceral leishmaniasis, especially in southern Europe. This is due to primarily tosharing of needles and syringes. This route of infection has been extending to the Nordic countries.
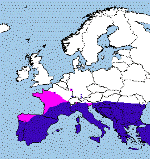
Leishmania is largely distributed in tropical and subtropical areas, extending from Central America to the Mediterranean countries, Africa, Central Asia, India, and China. Leishmaniasis is considered the second most important protozoal disease in the world, with its incidence being lower than malaria only.
In humans, visceral leishmaniasis affects mainly children and immunosuppressed individuals, and it is characterized by long periods of irregular fever, weakness, weight loss, pancytopenia, hepato- and splenomegaly, hypergammaglobulinemia, and hypoalbuminemiawhich progresses to chronicity or death in the absence of adequate treatment.
Several species of canids (dogs, foxes, and coyotes), marsupials, and rodents have been incriminated as reservoirs in endemic areas. Dogs play an essential role for maintenance of the disease in endemic areas where they act as domestic reservoir and source of infection for the Phlebotomus and consequently human infections.
Clinical Signs
Clinical leishmaniasis is a spectrum of diseases with the host immune response determining the type of syndrome which develops, and the clinical picture is one of slow, progressive evolution of illness. Syndromes range from mild self-limiting local skin lesions to systemic fatal diseases. Visceral leishmaniasis starts as a cutaneous lesion and the infection spreads systemically. Organs particularly affected are spleen, liver and bone marrow.
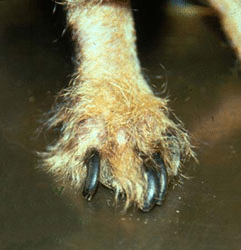
The main dermatologic signs of visceral leishmaniasis consist of emaciation, keratitis, scaling of skin, seborrhea, onicogripfosis, ulceration, and alopecia, which is often symmetrical in distribution. Some heavily infected dogs don´t have any clinical signs. Frequently there is also a discrepancy between the intensity of infection and the clinical condition in dogs.
With the visceral form, the disease can be severe, causing clinical signs in many organs. There are: extreme weakness, wasting, diarrhea, epistaxis, lameness, anemia, renal failure, edema of the feet, dermal ulceration, eye inflammation leading to blindness, lymphadenopathy and hepatosplenomegaly.
Body temperature may fluctuate but is usually normal or subnormal. Immunosuppression may promote the occurrence of concomitant infections, hence the clinical picture may be complicated by demodicosis, pyoderma or pneumonia.
Diagnosis
The diagnosis of leishmania is difficult because the clinical signs are variable, the histopathology is non-specific, and the microscopic lesions are also observed in other immune-mediated diseases and infections.
The diagnosis is divided into three main catagories:
• Parasitologic diagnosis
• Immunologic diagnosis
• Molecular methods (amplification of parasite DNA)
Parasitologic DiagnosisThis is the simplest and most commonly performed procedure. The diagnosis is based on cytologic or histopathologic observation of amastigotes in giemsa stained smears of bone marrow or lymph node aspirates. Immunoperoxidase staining improves the detection of amastigotes in the infected tissues. This method is highly specific and cheap, but it has a low sensitivity.
Canine Bone Marrow cytology-Leishmania organisms concentrated within macrophages
Canine Integuement histopathology- immunoperoxidase stained amastigotes Immunologic Diagnosis
This is the detection of antibodies (mainly IgG and especially IgG1) against Leishmania parasites or a specific cell-mediated response. The four main serological tests performed are IFAT, ELISA, DAT, and Western Blot. IFAT is considered the gold standard of tests, it has a high specificity and high sensitivity. IFAT uses the whole organism, which gives more repeatable and reliable results, rather than those using soluble Ag, such as complement fixation. ELISA is more sensitive, but less precise than IFAT. ELISA also cross reacts with Trypanosoma cruzi and Babesia.
Molecular Methods (amplification/detection of Leishmania DNA)
PCR uses primers from the rRNA gene to identify parasites from a variety of samples, including canine and human bone marrow, lymph nodes, skin biopsies, and heparinized whole blood. PCR is useful in the diagnosis of Leishmania, for follow-up patients pre and post treatment, and for the identification of parasitic species. PCR is a very sensitive test, but is not always available.
Differential Diagnosis
The clinical presentation of Leishmaniasis can vary greatly ranging from asymptomatic to a severe wasting disease. A differential diagnosis must be formulated for each individual case.
Treatment, Control and Prevention
The canine visceral leishmaniasis (Kala-azar) has been treated by European veterinarians for many years. In Brazil, therapeutic protocols for dogs have been evaluated over the last 4 years, but treatment of infected dogs is not recommended due to the potential risk to public health. The control policy adopted by the Brazilian Ministry of Health includes early treatment of human infections, strategic use of insecticides, and elimination of seropositive dogs.
The currently used protocol was proposed by Ferrer in 1997. The drugs most commonly used are: Meglumine Antimoniate (Glucantime®) associated with Allopurinol (Zyloric®), Aminosidine (Gabbriomycin®), and, recently, Amphotericin B (Fungizone®). All these drugs require a multiple dose regimen, and this will depend on the patient’s condition and owner cooperation.
It issuggested that maintenence treatment should be kept with allopurinol, because it is not possible to ensure that dogs will not relapse if treatment is discontinued.
The use of collars containing insecticides, shampoos or sprays effective to protect dogs from sandfly bites must be continuously used for all patients under treatment.
The vector control is one of the most important aspects of disease control.. The sandfly is vulnerable to the same insecticides as the malaria vector.
Residual insecticide spraying of houses and animal shelters will have an impact on transmission only when the vector is restricted to the intra- and peridomestic area.